Our research
Cytotoxic T Lymphocytes
The Lieberman lab studies innate and adaptive immune responses to infection and cancer as well as the molecular basis of killer cell-mediated cytotoxicity. They described the first caspase-independent programmed cell death pathway (activated by granzyme A) and made major contributions to understanding the function of killer cell pore-forming proteins. The lab identified novel mechanisms of mitochondrial and DNA damage and RNA degradation activated by killer lymphocytes. Other work contributed to understanding how cytotoxic T lymphocyte function is regulated, particularly in chronic infections. The lab developed a new model for how perforin delivers the cytolytic proteases (granzymes) into target cells by activating the plasma membrane repair response. The lab was the first to describe CD8 T cell exhaustion in humans, which is the basis for current checkpoint blockade therapies for treating cancer.
Recently, the lab described a new killer cell response to intracellular infection by bacteria, fungi and parasites in which perforin and another killer cell pore-forming protein (granulysin) deliver the granzymes into intracellular microbes to trigger their programmed cell death (“microptosis”). They also uncovered a new mechanism by which NK cells can kill intracellular bacteria without killing the host cell by transferring granulysin via nanotubes.
Pyroptosis
The Lieberman lab identified the mechanism behind gasdermin-mediated inflammatory death (pyroptosis) triggered by innate immune recognition of pathogens and danger signals. They study how pyroptosis is triggered and regulated. Recent work identified important roles for pyroptosis in SARS-CoV-2, Yersinia, and Group A streptococcal infections, in immune control of cancer and neurodegeneration.
They helped solve the structure of gasdermin pores and identify by a high throughput screen a potent GSDMD/pyroptosis inhibitor that protects mice from sepsis. More recently they showed that another gasdermin (GSDME) is a potent tumor suppressor. GSDME is not expressed or is mutated in most cancers. When GSDME is expressed in tumor cells, it ignites a potent immune response in which ordinarily dysfunctional tumor-infiltrating lymphocytes become potent anti-tumor killers. They also showed that granzyme B in killer cells activates pyroptosis in target cells that express GSDME by cleaving GSDME.
Viral infection
The Lieberman lab recently found that monocytes and macrophages are infected with SARS-CoV-2 and their infection triggers the assembly of inflammasomes, pyroptosis and release and processing of IL-1 family cytokines. Biomarkers of pyroptosis are increased in the blood of COVID patients and their increase correlates with COVID-19 disease severity. Infection of monocytes is mediated by Fc receptor uptake of antibody-coated virions, while infection of lung macrophages is mediated by both ACE2 and antibody-dependent FcR uptake. They are currently continuing this work to better understand antibody-mediated infection of monocytes, which do not express ACE2, and how their infection triggers inflammasome activation and pyroptosis and the features of monocytes and antibodies that facilitate infection.
Much of their viral work has been about HIV, for which the lab characterized the properties of HIV epitopes and used them to develop and test adoptive immunotherapy using ex vivo expanded antiviral CD8 T cells at a time when HIV antiviral drugs were not yet available. When T cell therapy didn’t work as well as expected, Dr. Lieberman discovered CD8 T cell dysfunction/exhaustion in humans, and also, developed strategies for RNA-based gene knockdown therapeutics for HSV and HIV. Dr. Lieberman published the first study to describe how HIV-infected cells avoid triggering innate immunity and an interferon response by harnessing Trex1, a cytosolic DNase to degrade reverse transcripts that would otherwise trigger the cGAS/STING pathway. Other work has examined the immune response to HSV, EBV, CMV, KSHV, and ZIKV.
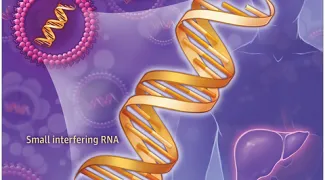
RNA interference
The laboratory has also been in the forefront of developing RNAi-based therapeutics and using RNAi for genome-wide screening. They were the first to show that siRNAs could be used to treat disease in vivo and to develop cell-targeted RNAs that knockdown gene expression in vivo in immune cells and cancer. They developed strategies for cell-specific delivery of siRNAs to tissues outside the liver that knockdown gene expression in vivo in T lymphocytes and cancer cells. They are currently investigating the use of aptamer-linked siRNAs for cancer immune therapy. They also investigate the intracellular trafficking of siRNA therapeutics and the role of microRNAs and lncRNAs in regulating cancer.
The Lieberman Lab is part of the Program in Cellular and Molecular Medicine at Boston Children's Hospital. The lab is affiliated with Harvard Medical School graduate programs in Biological and Biomedical Sciences, Immunology and Virology, and the Harvard Medical School Department of Genetics

The Lieberman Lab is part of the Program in Cellular and Molecular Medicine at Boston Children's Hospital. The lab is affiliated with Harvard Medical School Immunology Program, Harvard Genetic Program, and Harvard Stem Cell Institute.
